Over the last years, more and more tau-treatments are being assessed in mouse models for their potential to treat Alzheimer’s disease. One of the key challenges with many of the tau mouse models is the need for large numbers of animals due to their intrinsic variability.
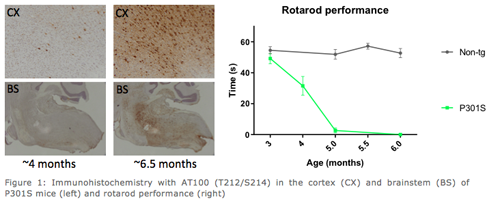
reMYND has complemented its portfolio of tau-models, which includes the well-characterized proprietary Tau.P301L model, with the Tau.P301S model. The Tau.P301S model embodies an aggressive, age-dependent tau-o-pathy, with manageable inter-mouse variability, a strong behaviour-pathology correlation and apparentforebrain pathology.
The progressive Tau hyper-phosphorylation and tangles with age (AT8, AT100), the inflammatory response (microgliosis, astrogliosis) and a strong correlation of Tau pathology with the behavioural phenotype (as measured by hind limb clasping and rotarod) make this model also ideal for testing efficacy of Tau-focused treatments. Several small molecule and immunization approaches have provided pivotal proof-of-concept data in the Tau.P301S transgenic model.
In a recent paper published in PLOS ONE, Ozelik and colleagues demonstrate a profound effect of short and long-term rapamycin treatments on various pathological read-outs in the brainstem and cortex of Tau.P301S mice (AT8, AT100, GFAP, …). In another paper published in the Journal of Biological Chemistry, Chai and coworkers show that short term passive immunization with Tau-directed antibodies lead to a preservation of motor function and strong effects on Tau pathology in the Tau.P301S model.